AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Periodic table molar mass of LiCl3/28/2024 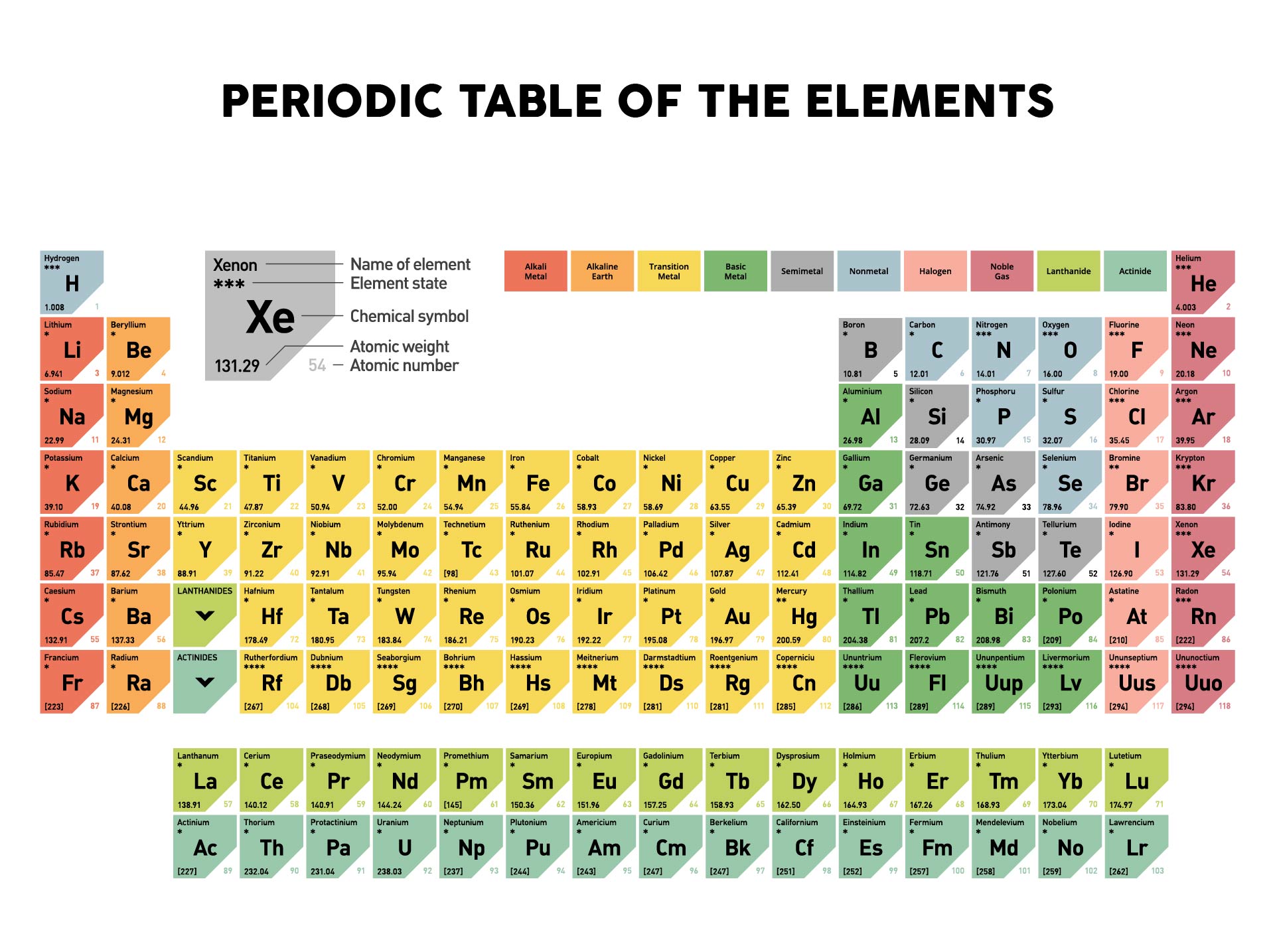 
Question 19 (0.125 points) Siu Whal is the formula mass of ammonia? Just report the Formula from the textbook Use the Periodic Table in Appendix A of your textbook and report your answer in units of "g/mol". Add them together: add the results from step 3 to get the total molar mass of the compound. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. Question 18 (0.125 points) How many grams of zinc are in 5.85 moles of zinc? Use the Periodic Table in Appendix A of your textbook and report your answer in units of "g". The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. The molar mass and molecular weight of LiCl is 42.394. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Question 17 (0.125 points) What is the percent chlorine (by mass) in lithium chloride? Use the Periodic Table in Appendix A of your textbook and report your answer in units of "%".
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
